Prognosis Kanker Ovarium Stadium Lanjut dalam Perspektif Biomarker Molekular: Kajian Ekspresi CD117, CD133, CD24, CD44, dan ALDH
Keywords:
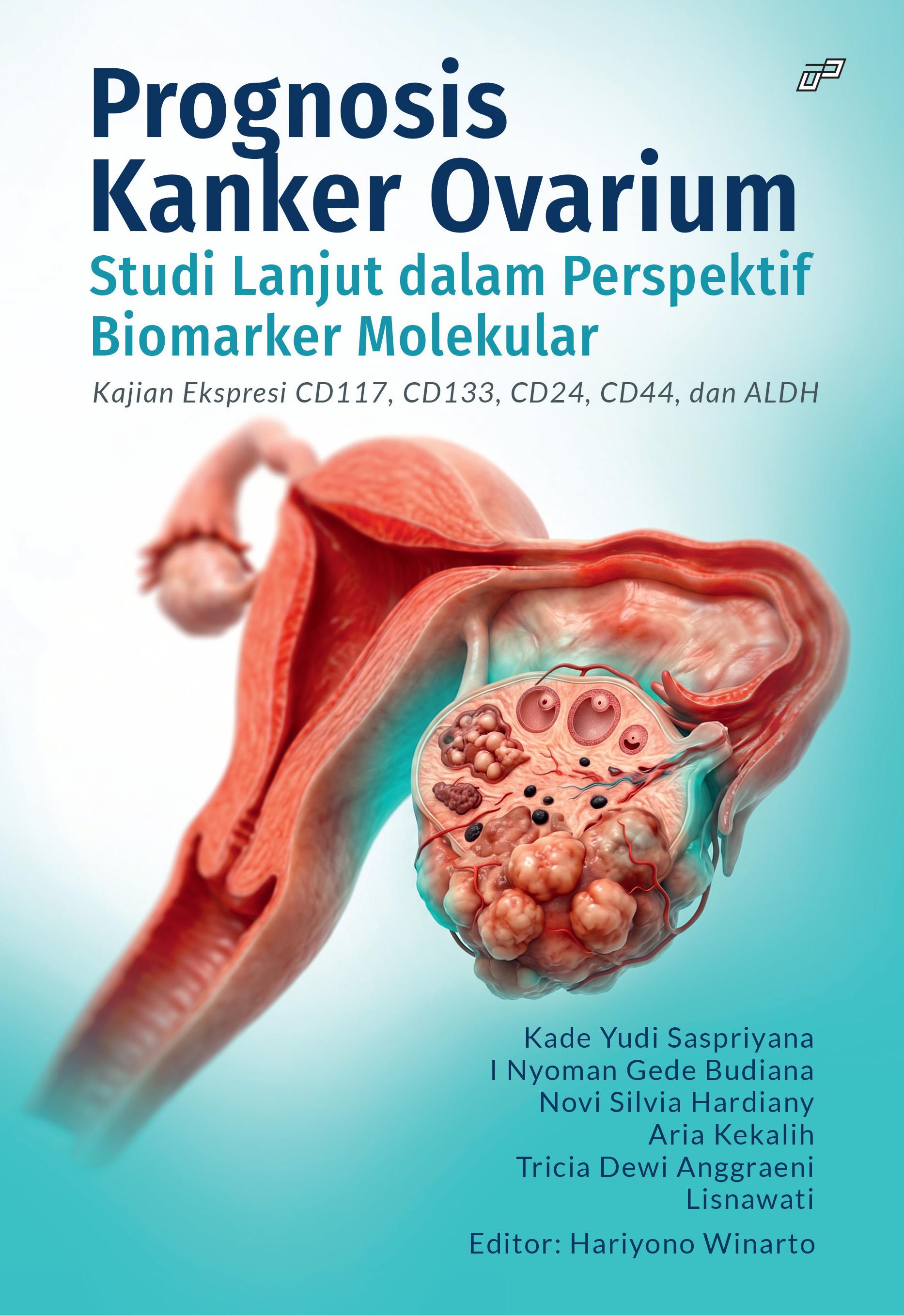
Kanker Ovarium Tipe Epitel, Sel Punca Kanker, CD117, CD133, CD24, CD44, ALDH, Progression Free SurvivalSynopsis
Prognosis Kanker Ovarium dalam Perspektif Biomarker Molekuler: Kajian Ekspresi CD117, CD133, CD24, CD44, dan ALDH merupakan monografi ilmiah yang membahas peran biomarker molekuler dalam memahami patogenesis dan prognosis kanker ovarium epitel stadium lanjut. Buku ini menyoroti pentingnya konsep cancer stem cell (CSC) atau sel punca kanker yang diyakini berperan dalam pertumbuhan tumor, resistensi terapi, kekambuhan penyakit, serta luaran klinis pasien.
Melalui pendekatan ilmiah yang sistematis, buku ini mengulas berbagai aspek penting, mulai dari epidemiologi kanker ovarium, konsep dasar sel punca kanker, hingga kajian ekspresi biomarker CSC seperti CD117, CD133, CD24, CD44, dan ALDH. Pembahasan juga mencakup metode penelitian berbasis imunohistokimia serta analisis statistik yang digunakan untuk menilai hubungan antara ekspresi biomarker dengan prognosis dan kemungkinan kekambuhan penyakit.
Dengan memadukan kajian teoretis dan analisis penelitian, buku ini memberikan gambaran yang komprehensif mengenai potensi biomarker molekuler dalam mendukung penentuan prognosis dan pengembangan terapi target di masa depan. Buku ini ditujukan bagi dokter spesialis, residen, mahasiswa kedokteran, peneliti biomedis, serta tenaga kesehatan yang memiliki minat dalam bidang onkologi ginekologi.
Downloads
References
Purbadi S, Saspriyana KY. Primary debulking surgery of advanced epithelial ovarian cancer in developing countries: challenges and expectations. Eur J Gynaecol Oncol. 2021;42(1):26.
Longo DL, Young RC. The natural history and treatment of ovarian cancer. Annu Rev Med. 1981;32:475–90.
Kurta ML, Edwards RP, Moysich KB, McDonough K, Bertolet M, Weissfeld JL, et al. Prognosis and conditional disease-free survival among patients with ovarian cancer. J Clin Oncol. 2014;32(36):4102–15.
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–49.
Pangribowo S. Beban kanker di Indonesia. Jakarta: Pusat Data dan Informasi Kesehatan Kementerian Kesehatan RI; 2019. p. 1–16.
Fitriani N, Sutrisna B, Pelupessy NU. Survival analysis of ovarian cancer patients based on age at Dr. Wahidin Sudirohusodo Hospital Makassar, 2014–2018. Indian J Public Health Res Dev. 2020;11(6):1397-402.
Mulawardhana P, Yuliati I, Sudiana K. Ekspresi CD44 sebagai faktor prognostik kekambuhan pada kanker ovarium tipe epitel stadium III. Indones J Cancer. 2017;11(3):119–29.
Budiana ING, Prayudi PKA, Saspriyana KY, Darmayasa IM, Wiradnyana AAG, Suwiyoga K. Characteristics of ovarian malignancy in Bali Province, Indonesia. Med J Malaysia. 2021;76(3):326.
Webb PM, Jordan SJ. Epidemiology of epithelial ovarian cancer. Best Pract Res Clin Obstet Gynaecol. 2017;41:1–12.
Zhang XY, Zhang PY. Recent perspectives of epithelial ovarian carcinoma. Oncol Lett. 2016;12(5):3055–8.
Shabir S, Gill PK. Global scenario on ovarian cancer: dynamics, relative survival, treatment, and epidemiology. Adesh Univ J Med Sci Res. 2020;2(1):17–25.
Prat J. Ovarian carcinomas: five distinct diseases with different origins, genetic alterations, and clinicopathological features. Virchows Arch. 2012;460(3):237–49.
Kroeger PT Jr, Drapkin R. Pathogenesis and heterogeneity of ovarian cancer. Curr Opin Obstet Gynecol. 2017;29(1):26–34.
Armstrong DK, Alvarez RD, Backes FJ, Bakkum-Gamez JN, Barroilhet L, Behbakht K, et al. NCCN guidelines insights: ovarian cancer, version 3.2022. J Natl Compr Canc Netw. 2022;20(9):972–80.
Reid BM, Permuth JB, Sellers TA. Epidemiology of ovarian cancer: a review. Cancer Biol Med. 2017;14(1):9–32.
Paoletti X, Lewsley LA, Daniele G, Cook A, Yanaihara N, Tinker A, et al. Assessment of progression-free survival as a surrogate end point of overall survival in first-line treatment of ovarian cancer: a systematic review and meta-analysis. JAMA Netw Open. 2020;3(1):e19189
Oza AM, Castonguay V, Tsoref D, Diaz-Padilla I, Karakasis K, Mackay H, et al. Progression-free survival in advanced ovarian cancer: a Canadian review and expert panel perspective. Curr Oncol. 2021;18(12):S20–7.
Chase DM, Mahajan A, Scott DA, Hawkins N, Kalilani L. Correlation between progression-free survival and overall survival in patients with ovarian cancer after cytoreductive surgery: a systematic literature review. Int J Gynecol Cancer. 2023;33(10):1602–11.
Anggraeni TD, Thiono J, Pratiwi IW, Hariadi A, Kispa TD, Kekalih A, et al. The presence of stem cells in ovarian cancer: a review. Bali Med J. 2023;12(1):1001–8.
Motohara T, Yoshida GJ, Katabuchi H. The hallmarks of ovarian cancer stem cells and niches: exploring their harmonious interplay in therapy resistance. Semin Cancer Biol. 2021;77:182–93.
Doubeni CA, Doubeni AR, Myers AE. Diagnosis and management of ovarian cancer. Am Fam Physician. 2016;93(11):937–44.
Khonsa O, Nuranna L, Sutrisna B. Kesintasan Pasien Karsinoma Ovarium dan Faktor-faktor yang Mempengaruhinya di RSUPN Dr. Cipto Mangunkusumo Jakarta (Pemantauan 5 tahun). Maj Obstet Ginekol Indonesia. 2007;31(2):66-72.
Kartini K, Pawitan JA. Sel punca kanker (cancer stem cells): sebuah tinjauan pustaka. Maj Patol Indones. 2018;27(3):36–43.
Fulawka L, Donizy P, Halon A. Cancer stem cells—the current status of an old concept: literature review and clinical approaches. Biol Res. 2014;47(1):66.
Królewska-Daszczyńska P, Wendlocha D, Smycz-Kubańska M, Stępień S, Mielczarek-Palacz A. Cancer stem cell markers in ovarian cancer: clinical and therapeutic significance. Oncol Lett. 2022;24(6):465.
Klemba A, Purzycka-Olewiecka JK, Wcisło G, Czarnecka AM, Lewicki S, Lesyng B, et al. Surface markers of cancer stem-like cells of ovarian cancer and their clinical relevance. Contemp Oncol. 2018;22(1):48–55.
Parte SC, Batra SK, Kakar SS. Characterization of stem cell and cancer stem cell populations in ovary and ovarian tumors. J Ovarian Res. 2018;11(1):69.
Phi LTH, Sari IN, Yang YG, Lee SH, Jun N, Kim KS, et al. Cancer stem cells (CSCs) in drug resistance and their therapeutic implications in cancer treatment. Stem Cells Int. 2018;2018:5416923.
Lin J, Ding D. The prognostic role of the cancer stem cell marker CD44 in ovarian cancer: a meta-analysis. Cancer Cell Int. 2017;17:8.
Tao Y, Li H, Huang R, Mo D, Zeng T, Fang M, et al. Clinicopathological and prognostic significance of cancer stem cell markers in ovarian cancer patients: evidence from 52 studies. Cell Physiol Biochem. 2018;46(4):1716–26.
Meng E, Mitra A, Tripathi K, Finan MA, Scalici J, McClellan S, et al. ALDH1A1 maintains ovarian cancer stem cell-like properties by altered regulation of cell cycle checkpoint and DNA repair network signaling. PLoS One. 2014;9(9):e107142.
Mizuno T, Suzuki N, Makino H, Furui T, Morii E, Aoki H, et al. Cancer stem-like cells of ovarian clear cell carcinoma are enriched in the ALDH-high population associated with an accelerated scavenging system in reactive oxygen species. Gynecol Oncol. 2015;137(2):299–305.
Roy M, Connor J, Al-Niaimi A, Rose SL, Mahajan A. Aldehyde dehydrogenase 1A1 expression by immunohistochemistry is associated with chemo-refractoriness in patients with high-grade ovarian serous carcinoma. Hum Pathol. 2018;73:1–6.
Peitzsch C, Kurth I, Kunz-Schughart L, Baumann M, Dubrovska A. Discovery of the cancer stem cell related determinants of radioresistance. Radiother Oncol. 2013;108(3):378–87.
Ishimoto T, Nagano O, Yae T, Tamada M, Motohara T, Oshima H, et al. CD44 variant regulates redox status in cancer cells by stabilizing the xCT subunit of system Xc− and thereby promotes tumor growth. Cancer Cell. 2011;19(3):387–400.
Li W, Ma H, Zhang J, Zhu L, Wang C, Yang Y. Unraveling the roles of CD44/CD24 and ALDH1 as cancer stem cell markers in tumorigenesis and metastasis. Sci Rep. 2017;7:13856.
Meng E, Long B, Sullivan P, McClellan S, Finan MA, Reed E, et al. CD44+/CD24− ovarian cancer cells demonstrate cancer stem cell properties and correlate to survival. Clin Exp Metastasis. 2012;29(8):939–48.
Varier L, Sundaram SM, Gamit N, Warrier S. An overview of ovarian cancer: the role of cancer stem cells in chemoresistance and a precision medicine approach targeting the Wnt pathway with the antagonist SFRP4. Cancers (Basel). 2023;15(4):1275.
Conic I, Stanojevic Z, Jankovic Velickovic L, Stojnev S, Ristic Petrovic A, Krstic M, et al. Epithelial ovarian cancer with CD117 phenotype is highly aggressive and resistant to chemotherapy. J Obstet Gynaecol Res. 2015;41(10):1630–7.
Zhang J, Guo X, Chang DY, Rosen DG, Mercado-Uribe I, Liu J. CD133 expression associated with poor prognosis in ovarian cancer. Mod Pathol. 2012;25(3):456–64.
Ruengwanichayakun P. Histochemical scoring assessment (H-score). Asian Arch Pathol. 2021;3:13–14.
Manek S. The histological value of chemotherapy response score in advanced ovarian cancer—histopathological challenges. Gynecol Pelvic Med. 2023;6:1–7.
Torre LA, Trabert B, DeSantis CE, Miller KD, Samimi G, Runowicz CD, et al. Ovarian cancer statistics, 2018. CA Cancer J Clin. 2018;68(4):284–96.
Momenimovahed Z, Tiznobaik A, Taheri S, Salehiniya H. Ovarian cancer in the world: epidemiology and risk factors. Int J Womens Health. 2019;11:287–99.
Chan JK, Urban R, Cheung MK, Osann K, Husain A, Teng NN, et al. Ovarian cancer in younger vs older women: a population-based analysis. Br J Cancer. 2006;95(10):1314–20.
Zheng G, Yu H, Kanerva A, Försti A, Sundquist K, Hemminki K. Familial risks of ovarian cancer by age at diagnosis, proband type and histology. PLoS One. 2018;13(10):e0205000.
Arora N, Talhouk A, McAlpine JN, Law MR, Hanley GE. Long-term mortality among women with epithelial ovarian cancer: a population-based study in British Columbia, Canada. BMC Cancer. 2018;18(1):1039.
Ørskov M, Iachina M, Guldberg R, Mogensen O, Nørgård BM. Predictors of mortality within 1 year after primary ovarian cancer surgery: a nationwide cohort study. BMJ Open. 2016;6(4):e010123.
Præstegaard C, Kjaer SK, Nielsen TS, Jensen SM, Webb PM, Nagle CM, et al. The association between socioeconomic status and tumour stage at diagnosis of ovarian cancer: a pooled analysis of 18 case-control studies. Cancer Epidemiol. 2016;41:71–9.
Alberg AJ, Moorman PG, Crankshaw S, Wang F, Bandera EV, Barnholtz-Sloan JS, et al. Socioeconomic status in relation to the risk of ovarian cancer in African-American women: a population-based case-control study. Am J Epidemiol. 2016;184(4):274–83.
Kim SJ, Rosen B, Fan I, Ivanova A, McLaughlin JR, Risch H, et al. Epidemiologic factors that predict long-term survival following a diagnosis of epithelial ovarian cancer. Br J Cancer. 2017;116(7):964–71.
Fujita M, Tase T, Kakugawa Y, Hoshi S, Nishino Y, Nagase S, et al. Smoking, earlier menarche and low parity as independent risk factors for gynecologic cancers in Japanese: a case-control study. Tohoku J Exp Med. 2008;216(4):297–307.
Weiderpass E, Sandin S, Inoue M, Shimazu T, Iwasaki M, Sasazuki S, et al. Risk factors for epithelial ovarian cancer in Japan: results from the Japan Public Health Center-based prospective study cohort. Int J Oncol. 2012;40(1):21–30.
Risch HA, Marrett LD, Jain M, Howe GR. Differences in risk factors for epithelial ovarian cancer by histologic type: results of a case-control study. Am J Epidemiol. 1996;144(4):363–72.
Merritt MA, Green AC, Nagle CM, Webb PM; Australian Cancer Study (Ovarian Cancer) and Australian Ovarian Cancer Study Group. Talc use, body powder exposure, and risk of ovarian cancer. Cancer Epidemiol Biomarkers Prev. 2008;17(5):1212–24.
Chiang AJ, Chang C, Huang CH, Huang WC, Kan YY, Chen J. Risk factors in progression from endometriosis to ovarian cancer: a cohort study based on medical insurance data. J Gynecol Oncol. 2018;29(3):e28.
Pavone ME, Lyttle BM. Endometriosis and ovarian cancer: links, risks, and challenges faced. Int J Womens Health. 2015;7:663–72.
Hartge P, Hayes R, Reding D, Sherman ME, Prorok P, Schiffman M, et al. Complex ovarian cysts in postmenopausal women are not associated with ovarian cancer risk factors: preliminary data from the Prostate, Lung, Colon, and Ovarian Cancer Screening Trial. Am J Obstet Gynecol. 2000;183(5):1232–7.
Sieh W, Salvador S, McGuire V, Weber RP, Terry KL, Rossing MA, et al. Tubal ligation and risk of ovarian cancer subtypes: a pooled analysis of case-control studies. Int J Epidemiol. 2013;42(2):579–89.
Tsilidis KK, Allen NE, Key TJ, Dossus L, Lukanova A, Bakken K, et al. Oral contraceptive use and reproductive factors and risk of ovarian cancer in the European Prospective Investigation into Cancer and Nutrition. Br J Cancer. 2011;105(9):1436–42.
Royar J, Becher H, Chang-Claude J. Low-dose oral contraceptives: protective effect on ovarian cancer risk. Int J Cancer. 2001;95(6):370–4.
Moorman PG, Calingaert B, Palmieri RT, Iversen ES, Bentley RC, Halabi S, et al. Hormonal risk factors for ovarian cancer in premenopausal and postmenopausal women. Am J Epidemiol. 2008;167(9):1059–69.
Rossing MA, Cushing-Haugen KL, Wicklund KG, Doherty JA, Weiss NS. Menopausal hormone therapy and risk of epithelial ovarian cancer. Cancer Epidemiol Biomarkers Prev. 2007;16(12):2548–56.
Lacey JV Jr, Mink PJ, Lubin JH, Sherman ME, Troisi R, Hartge P, et al. Menopausal hormone replacement therapy and risk of ovarian cancer. JAMA. 2002;288(3):334–41.
Mørch LS, Løkkegaard E, Andreasen AH, Krüger-Kjær S, Lidegaard Ø. Hormone therapy and ovarian cancer. JAMA. 2009;302(3):298–305.
Reigstad MM, Storeng R, Myklebust TÅ, Oldereid NB, Omland AK, Robsahm TE, et al. Cancer risk in women treated with fertility drugs according to parity status—a registry-based cohort study. Cancer Epidemiol Biomarkers Prev. 2017;26(6):953–62.
Shushan A, Paltiel O, Iscovich J, Elchalal U, Peretz T, Schenker JG. Human menopausal gonadotropin and the risk of epithelial ovarian cancer. Fertil Steril. 1996;65(1):13–18.
Calderon-Margalit R, Friedlander Y, Yanetz R, Kleinhaus K, Perrin MC, Manor O, et al. Cancer risk after exposure to treatments for ovulation induction. Am J Epidemiol. 2009;169(3):365–75.
Kazerouni N, Greene MH, Lacey JV Jr, Mink PJ, Schairer C. Family history of breast cancer as a risk factor for ovarian cancer in a prospective study. Cancer. 2006;107(5):1075–83.
Mori M, Harabuchi I, Miyake H, Casagrande JT, Henderson BE, Ross RK. Reproductive, genetic, and dietary risk factors for ovarian cancer. Am J Epidemiol. 1988;128(4):771–7.
Walsh T, Casadei S, Lee MK, Pennil CC, Nord AS, Thornton AM, et al. Mutations in 12 genes for inherited ovarian, fallopian tube, and peritoneal carcinoma identified by massively parallel sequencing. Proc Natl Acad Sci U S A. 2011;108(44):18032–7.
Toss A, Tomasello C, Razzaboni E, Contu G, Grandi G, Cagnacci A, et al. Hereditary ovarian cancer: not only BRCA1 and BRCA2 genes. Biomed Res Int. 2015;2015:341723.
Andrews L, Mutch DG. Hereditary ovarian cancer and risk reduction. Best Pract Res Clin Obstet Gynaecol. 2017;41:31–48.
Kotsopoulos J, Gronwald J, Karlan B, Rosen B, Huzarski T, Moller P, et al. Age-specific ovarian cancer risks among women with a BRCA1 or BRCA2 mutation. Gynecol Oncol. 2018;150(1):85–91.
Kauff ND, Satagopan JM, Robson ME, Scheuer L, Hensley M, Hudis CA, et al. Risk-reducing salpingo-oophorectomy in women with a BRCA1 or BRCA2 mutation. N Engl J Med. 2002;346(21):1609–15.
Iordache PD, Mates D, Gunnarsson B, Eggertsson HP, Sulem P, Benonisdottir S, et al. Identification of Lynch syndrome risk variants in the Romanian population. J Cell Mol Med. 2018;22(12):6068–76.
Nakamura K, Banno K, Yanokura M, Iida M, Adachi M, Masuda K, et al. Features of ovarian cancer in Lynch syndrome. Mol Clin Oncol. 2014;2(6):909–16.
Lu KH, Daniels M. Endometrial and ovarian cancer in women with Lynch syndrome: update in screening and prevention. Fam Cancer. 2013;12(2):273–7.
Ong JS, Cuellar-Partida G, Lu Y, Australian Ovarian Cancer Study, Fasching PA, Hein A, et al. Association of vitamin D levels and risk of ovarian cancer: a Mendelian randomization study. Int J Epidemiol. 2016;45(5):1619–30.
Bandera EV, Lee VS, Qin B, Rodriguez-Rodriguez L, Powell CB, Kushi LH. Impact of body mass index on ovarian cancer survival varies by stage. Br J Cancer. 2017;117(2):282–9.
Delort L, Kwiatkowski F, Chalabi N, Satih S, Bignon YJ, Bernard-Gallon DJ. Central adiposity as a major risk factor of ovarian cancer. Anticancer Res. 2009;29(12):5229–34.
Beehler GP, Sekhon M, Baker JA, Teter BE, McCann SE, Rodabaugh KJ, et al. Risk of ovarian cancer associated with BMI varies by menopausal status. J Nutr. 2006;136(11):2881–6.
Cottreau CM, Ness RB, Kriska AM. Physical activity and reduced risk of ovarian cancer. Obstet Gynecol. 2000;96(4):609–14.
Anderson JP, Ross JA, Folsom AR. Anthropometric variables, physical activity, and incidence of ovarian cancer: the Iowa Women’s Health Study. Cancer. 2004;100(7):1515–21.
Goodman MT, Wu AH, Tung KH, McDuffie K, Kolonel LN, Nomura AM, et al. Association of dairy products, lactose, and calcium with the risk of ovarian cancer. Am J Epidemiol. 2002;156(2):148–57.
Schouten LJ, Zeegers MP, Goldbohm RA, van den Brandt PA. Alcohol and ovarian cancer risk: results from the Netherlands Cohort Study. Cancer Causes Control. 2004;15(2):201–9.
Kuper H, Titus-Ernstoff L, Harlow BL, Cramer DW. Population-based study of coffee, alcohol and tobacco use and risk of ovarian cancer. Int J Cancer. 2000;88(2):313–8.
Marchbanks PA, Wilson H, Bastos E, Cramer DW, Schildkraut JM, Peterson HB. Cigarette smoking and epithelial ovarian cancer by histologic type. Obstet Gynecol. 2000;95(2):255–60.
Su D, Pasalich M, Lee AH, Binns CW. Ovarian cancer risk is reduced by prolonged lactation: a case-control study in southern China. Am J Clin Nutr. 2013;97(2):354–9.
Tung KH, Goodman MT, Wu AH, McDuffie K, Wilkens LR, Kolonel LN, et al. Reproductive factors and epithelial ovarian cancer risk by histologic type: a multiethnic case-control study. Am J Epidemiol. 2003;158(7):629–38.
Berek JS, Kehoe ST, Kumar L, Friedlander M. Cancer of the ovary, fallopian tube, and peritoneum. Int J Gynecol Obstet. 2018;143(Suppl 2):59–78.
Ponnusamy MP, Batra SK. Ovarian cancer: emerging concept on cancer stem cells. J Ovarian Res. 2008;1(1):4.
Newton C, Murali K, Ahmad A, Hockings H, Graham R, Liberale V, et al. A multicentre retrospective cohort study of ovarian germ cell tumours: evidence for chemotherapy de-escalation and alignment of paediatric and adult practice. Eur J Cancer. 2019;113:19–27.
Dhitayoni IA, Budiana ING. Profil pasien kanker ovarium di Rumah Sakit Umum Pusat Sanglah Denpasar-Bali periode Juli 2013–Juni 2014. E-Jurnal Medika. 2017;6(3):1–9.
Matz M, Coleman MP, Sant M, Chirlaque MD, Visser O, Gore M, et al. The histology of ovarian cancer: worldwide distribution and implications for international survival comparisons (CONCORD-2). Gynecol Oncol. 2017;144(2):405–13.
Horta M, Cunha TM. Sex cord-stromal tumors of the ovary: a comprehensive review and update for radiologists. Diagn Interv Radiol. 2015;21(4):277–85.
Thrall MM, Paley P, Pizer E, Garcia R, Goff BA. Patterns of spread and recurrence of sex cord-stromal tumors of the ovary. Gynecol Oncol. 2011;122(2):242–5.
Javadi S, Ganeshan DM, Qayyum A, Iyer RB, Bhosale P. Ovarian cancer, the revised FIGO staging system, and the role of imaging. AJR Am J Roentgenol. 2016;206(6):1351–60.
Tarhriz V, Bandehpour M, Dastmalchi S, Ouladsahebmadarek E, Zarredar H, Eyvazi S. Overview of CD24 as a new molecular marker in ovarian cancer. J Cell Physiol. 2019;234(3):2134–42.
Trihandini I, Nurrika D. Analisis Ketahanan Hidup Lima Tahun Penderita Kanker Ovarium Epithelial di Rumah Sakit Kanker Dharmais Jakarta. Jurnal Kesehatan Masyarakat Nasional. 2010;5(3):139-45.
Shao C, Guo H, Chen L, Chen J, Wang L, Wang H. Prognostic factors and clinicopathologic characteristics of ovarian tumors with different histologic subtypes: a SEER database population study of 41,376 cases. Transl Cancer Res. 2023;12(8):1937–1950.
Andreou M, Kyprianidou M, Cortas C, Polycarpou I, Papamichael D, Kountourakis P, et al. Prognostic factors influencing survival in ovarian cancer patients: a 10-year retrospective study. Cancers (Basel). 2023;15(24):5710.
Ezzati M, Abdullah A, Shariftabrizi A, Hou J, Kopf M, Stedman JK, et al. Recent advancements in prognostic factors of epithelial ovarian carcinoma. Int Sch Res Notices. 2014;2014:953509.
Matsuo K, Sheridan TB, Yoshino K, Miyake T, Hew KE, Im DD, et al. Significance of lymphovascular space invasion in epithelial ovarian cancer. Cancer Med. 2012;1(2):156–164.
Yamashita YM, Yuan H, Cheng J, Hunt AJ. Polarity in stem cell division: asymmetric stem cell division in tissue homeostasis. Cold Spring Harb Perspect Biol. 2010;2(1):a001313.
Plaks V, Kong N, Werb Z. The cancer stem cell niche: how essential is the niche in regulating stemness of tumor cells? Cell Stem Cell. 2015;16(3):225–238.
Dick JE. Looking ahead in cancer stem cell research. Nat Biotechnol. 2009;27(1):44–46.
Rich JN. Cancer stem cells: understanding tumor hierarchy and heterogeneity. Medicine (Baltimore). 2016;95(1 Suppl):S2–S7.
Andriani F, Bertolini G, Facchinetti F, Baldoli E, Moro M, Casalini P, et al. Conversion to stem-cell state in response to microenvironmental cues is regulated by balance between epithelial and mesenchymal features in lung cancer cells. Mol Oncol. 2016;10(2):253–271.
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, et al. The epithelial–mesenchymal transition generates cells with properties of stem cells. Cell. 2008;133(4):704–715.
Xie M, Zhang L, He CS, Xu F, Liu JL, Hu ZH, et al. Activation of Notch-1 enhances epithelial–mesenchymal transition in gefitinib-acquired resistant lung cancer cells. J Cell Biochem. 2012;113(5):1501–1513.
Lo JF, Yu CC, Chiou SH, Huang CY, Jan CI, Lin SC, et al. The epithelial–mesenchymal transition mediator S100A4 maintains cancer-initiating cells in head and neck cancers. Cancer Res. 2011;71(5):1912–1923.
Siebzehnrubl FA, Silver DJ, Tugertimur B, Deleyrolle LP, Siebzehnrubl D, Sarkisian MR, et al. The ZEB1 pathway links glioblastoma initiation, invasion and chemoresistance. EMBO Mol Med. 2013;5(8):1196–1212.
Alisi A, Cho WC, Locatelli F, Fruci D. Multidrug resistance and cancer stem cells in neuroblastoma and hepatoblastoma. Int J Mol Sci. 2013;14(12):24706–24725.
Szotek PP, Pieretti-Vanmarcke R, Masiakos PT, Dinulescu DM, Connolly D, Foster R, et al. Ovarian cancer side population defines cells with stem cell-like characteristics and Müllerian inhibiting substance responsiveness. Proc Natl Acad Sci U S A. 2006;103(30):11154–11159.
Begicevic RR, Falasca M. ABC transporters in cancer stem cells: beyond chemoresistance. Int J Mol Sci. 2017;18(11):2362.
Bao B, Ali S, Ahmad A, Azmi AS, Li Y, Banerjee S, et al. Hypoxia-induced aggressiveness of pancreatic cancer cells is due to increased expression of VEGF, IL-6 and miR-21, which can be attenuated by CDF treatment. PLoS One. 2012;7(12):e50165.
Ng KP, Manjeri A, Lee KL, Huang W, Tan SY, Chuah CT, et al. Physiologic hypoxia promotes maintenance of CML stem cells despite effective BCR-ABL1 inhibition. Blood. 2014;123(21):3316–3326.
Murakami A, Takahashi F, Nurwidya F, Kobayashi I, Minakata K, Hashimoto M, et al. Hypoxia increases gefitinib-resistant lung cancer stem cells through activation of insulin-like growth factor 1 receptor. PLoS One. 2014;9(1):e86459.
Chau WK, Ip CK, Mak AS, Lai HC, Wong AS. c-Kit mediates chemoresistance and tumor-initiating capacity of ovarian cancer cells through activation of Wnt/β-catenin–ABCG2 signaling. Oncogene. 2013;32(22):2767–2779.
Abdullah LN, Chow EK. Mechanisms of chemoresistance in cancer stem cells. Clin Transl Med. 2013;2(1):3.
Bapat SA, Mali AM, Koppikar CB, Kurrey NK. Stem and progenitor-like cells contribute to the aggressive behavior of human epithelial ovarian cancer. Cancer Res. 2005;65(8):3025–3029.
Nowicki A, Kulus M, Wieczorkiewicz M, Pieńkowski W, Stefańska K, Skupin-Mrugalska P, et al. Ovarian cancer and cancer stem cells—cellular and molecular characteristics, signaling pathways, and usefulness as a diagnostic tool. Cancers (Basel). 2021;13(16):4178.
Parmar N, Gupta VK. Eradication of ovarian cancer stem cells in ovarian cancer using stem cell therapy. International Journal of Biotech Trends and Technology. 2021;11(2):15-24.
Foster BM, Zaidi D, Young TR, Mobley ME, Kerr BA. CD117/c-Kit in cancer stem cell-mediated progression and therapeutic resistance. Biomedicines. 2018;6(1):31.
Suster NK, Virant-Klun I. Presence and role of stem cells in ovarian cancer. World J Stem Cells. 2019;11(7):383–397.
Issar CR, Cunjian Y. Ovarian cancer stem cells in chemoresistance. Int J Sci Inventions Today. 2017;6(3):227–236.
Ferrandina G, Bonanno G, Pierelli L, Perillo A, Procoli A, Mariotti A, et al. Expression of CD133-1 and CD133-2 in ovarian cancer. Int J Gynecol Cancer. 2008;18(3):506–514.
Eyvazi S, Kazemi B, Bandehpour M, Dastmalchi S. Identification of a novel single-chain fragment variable antibody targeting CD24-expressing cancer cells. Immunol Lett. 2017;190:240–246.
Eyvazi S, Kazemi B, Dastmalchi S, Bandehpour M. Involvement of CD24 in multiple cancer-related pathways makes it an interesting new target for cancer therapy. Curr Cancer Drug Targets. 2018;18(4):328–336.
Nestl A, von Stein OD, Zatloukal K, Thies WG, Herrlich P, Hofmann M, et al. Gene expression patterns associated with the metastatic phenotype in rodent and human tumors. Cancer Res. 2001;61(4):1569–1577.
Nakamura K, Terai Y, Tanabe A, Ono YJ, Hayashi M, Maeda K, et al. CD24 expression is a marker for predicting clinical outcome and regulates epithelial–mesenchymal transition in ovarian cancer via both Akt and ERK pathways. Oncol Rep. 2017;37(6):3189–3200.
Baumann P, Cremers N, Kroese F, Orend G, Chiquet-Ehrismann R, Uede T, et al. CD24 expression causes the acquisition of multiple cellular properties associated with tumor growth and metastasis. Cancer Res. 2005;65(23):10783–10793.
Carroll MJ, Fogg KC, Patel HA, Krause HB, Mancha AS, Patankar MS, et al. Alternatively activated macrophages upregulate mesothelial expression of P-selectin to enhance adhesion of ovarian cancer cells. Cancer Res. 2018;78(13):3560–3573.
Abubaker K, Luwor RB, Zhu H, McNally O, Quinn MA, Burns CJ, Ahmed N. Inhibition of the JAK2/STAT3 pathway in ovarian cancer results in loss of cancer stem cell-like characteristics and reduced tumor burden. BMC Cancer. 2014;14:317.
Liang R, Chen X, Chen L, Wan F, Chen K, Sun Y, et al. STAT3 signaling in ovarian cancer: a potential therapeutic target. J Cancer. 2020;11(4):837–848.
Burgos-Ojeda D, Wu R, McLean K, Chen YC, Talpaz M, Yoon E, et al. CD24+ ovarian cancer cells are enriched for cancer-initiating cells and dependent on JAK2 signaling for growth and metastasis. Mol Cancer Ther. 2015;14(7):1717–1727.
Ponta H, Sherman L, Herrlich PA. CD44: from adhesion molecules to signalling regulators. Nat Rev Mol Cell Biol. 2003;4(1):33–45.
Sacks Suarez J, Gurler Main H, Muralidhar GG, Elfituri O, Xu HL, Kajdacsy-Balla AA, et al. CD44 regulates formation of spheroids and controls organ-specific metastatic colonization in epithelial ovarian carcinoma. Mol Cancer Res. 2019;17(9):1801–1814.
Yang B, Yan X, Liu L, Jiang C, Hou S. Overexpression of the cancer stem cell marker CD117 predicts poor prognosis in epithelial ovarian cancer patients: evidence from a meta-analysis. Onco Targets Ther. 2017;10:2951–2961.
Shnaider PV, Petrushanko IY, Aleshikova OI, Babaeva NA, Ashrafyan LA, Borovkova EI, et al. Expression level of CD117 (KIT) on ovarian cancer extracellular vesicles correlates with tumor aggressiveness. Front Cell Dev Biol. 2023;11:1057484.
Barzegar Behrooz A, Syahir A, Ahmad S. CD133: beyond a cancer stem cell biomarker. J Drug Target. 2019;27(3):257–269.
Ikram D, Masadah R, Nelwan BJ, Zainuddin AA, Ghaznawie M, Wahid S. CD133 acts as an essential marker in ovarian carcinogenesis. Asian Pac J Cancer Prev. 2021;22(11):3525–3531.
Zhou Q, Chen A, Song H, Tao J, Yang H, Zuo M. Prognostic value of cancer stem cell marker CD133 in ovarian cancer: a meta-analysis. Int J Clin Exp Med. 2015;8(3):3080–3089.
Baba T, Convery PA, Matsumura N, Whitaker RS, Kondoh E, Perry T, et al. Epigenetic regulation of CD133 and tumorigenicity of CD133+ ovarian cancer cells. Oncogene. 2009;28(2):209–218.
Chefetz I, Grimley E, Yang K, Hong L, Vinogradova EV, Suciu R, et al. A pan-ALDH1A inhibitor induces necroptosis in ovarian cancer stem-like cells. Cell Rep. 2019;26(11):3061–3075.
Tomita H, Tanaka K, Tanaka T, Hara A. Aldehyde dehydrogenase 1A1 in stem cells and cancer. Oncotarget. 2016;7(10):11018–11032.
Xia Y, Wei X, Gong H, Ni Y. Aldehyde dehydrogenase serves as a biomarker for worse survival profiles in ovarian cancer patients: an updated meta-analysis. BMC Womens Health. 2018;18(1):199.
Muralikrishnan V, Hurley TD, Nephew KP. Targeting aldehyde dehydrogenases to eliminate cancer stem cells in gynecologic malignancies. Cancers (Basel). 2020;12(4):961.
Steffensen KD, Alvero AB, Yang Y, Waldstrøm M, Hui P, Holmberg JC, et al. Prevalence of epithelial ovarian cancer stem cells correlates with recurrence in early-stage ovarian cancer. J Oncol. 2011;2011:620523.
Gao Y, Foster R, Yang X, Feng Y, Shen JK, Mankin HJ, et al. Up-regulation of CD44 in the development of metastasis, recurrence and drug resistance of ovarian cancer. Oncotarget. 2015;6(11):9313–9326.
Feharsal Y, Andrijono A, Singoprawiro CS, Lisnawati L, Pakasi TA, Putra AD, et al. Role of CD44 and CD24 expression on 2-year disease-free survival in patients with advanced epithelial ovarian carcinoma. Asian Pac J Cancer Prev. 2024;25(2):513–520.
Zhang C, Xu C, Gao X, Yao Q. Platinum-based drugs for cancer therapy and antitumor strategies. Theranostics. 2022;12(5):2115–2132.
Yang L, Xie HJ, Li YY, Wang X, Liu XX, Mai J. Molecular mechanisms of platinum-based chemotherapy resistance in ovarian cancer. Oncol Rep. 2022;47(4):1–11.
Abal M, Andreu JM, Barasoain I. Taxanes: microtubule and centrosome targets, and cell cycle-dependent mechanisms of action. Curr Cancer Drug Targets. 2003;3(3):193–203.
Maloney SM, Hoover CA, Morejon-Lasso LV, Prosperi JR. Mechanisms of taxane resistance. Cancers (Basel). 2020;12(11):3323.
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228–247.
Lemeshow S, Hosmer DW Jr, Klar J, Lwanga SK. Adequacy of sample size in health studies. Geneva: World Health Organization; 1990.
Tewari KS, Burger RA, Enserro D, Norquist BM, Swisher EM, Brady MF, et al. Final overall survival of a randomized trial of bevacizumab for primary treatment of ovarian cancer. J Clin Oncol. 2019;37(26):2317–2328.
González-Martín A, Harter P, Leary A, Lorusso D, Miller RE, Pothuri B, et al. Newly diagnosed and relapsed epithelial ovarian cancer: ESMO clinical practice guideline. Ann Oncol. 2023;34(10):833–848.
Pelupessy NU, Andrijono A, Sutrisna B, Harahap AR, Kanoko M, Nuranna L, et al. CD133, CD44, and ALDH1A1 as cancer stem cell markers and prognostic factors in epithelial ovarian cancer. Med J Indones. 2019;28(1):63–70.
Cullimore V, Gaitskell K, Newhouse R, Baxter K, Wood N, Fotopoulou C, et al. Advanced epithelial ovarian cancer in elderly patients. Int J Gynecol Cancer. 2025;35:102017.
Gu Y, Zhou G, Tang X, Shen F, Ding J, Hua K. The biological roles of CD24 in ovarian cancer: old story, but new tales. Front Immunol. 2023;14:1183285.
Nagare RP, Sneha S, Sidhanth C, Roopa S, Murhekar K, Shirley S, et al. Expression of cancer stem cell markers CD24, EPHA1 and CD9 and their correlation with clinical outcome in epithelial ovarian tumours. Cancer Biomark. 2020;28(3):397–406.
González-Martín A, Harter P, Leary A, Lorusso D, Miller RE, Pothuri B, et al. Newly diagnosed and relapsed epithelial ovarian cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Annals of Oncology. 2023 Oct 1;34(10):833-48.
Yassin, Mejalaa S, El Nashar A. The prognostic value of CD133 in ovarian surface epithelial tumors. Egypt J Hosp Med. 2022;86(1):891–897.
Gisina A, Kim Y, Yarygin K, Lupatov A. Can CD133 be regarded as a prognostic biomarker in oncology: pros and cons. Int J Mol Sci. 2023;24(24):17398.
Jovanović L, Šošić-Jurjević B, Ćirković A, Dragičević S, Filipović B, Milenković S, et al. Diagnostic potential of CD44, CD133, and VDR in epithelial ovarian tumors. Int J Mol Sci. 2025;26(8):3729.
Sihombing UHM, Andrijono A, Purwoto G, Gandamihardja S, Harahap AR, Rustamadji P, et al. CD44+/CD24− expression as predictors of ovarian cancer chemoresistance. J Egypt Natl Canc Inst. 2022;34(1):44.
Matuura H, Miyamoto M, Takano M, Soyama H, Aoyama T, Yoshikawa T, et al. Low expression of CD44 is an independent factor of poor prognosis in ovarian mucinous carcinoma. Anticancer Res. 2018;38(2):717–724.
Quaranta M, Laios A, Rogers C, Mavromatidou AI, Thangavelu A, Theophilou G, et al. Mapping the advanced-stage epithelial ovarian cancer landscape using large language models. J Clin Med. 2025;14(7):2223.
Al-Shami SA, Al-Kaabi MM, Mahdi AK, Al-Attar Z. Immunohistochemical expression of CD117 in borderline, low- and high-grade ovarian surface epithelial tumours. Malays J Pathol. 2023;45(2):229–238.
Ruscito I, Darb-Esfahani S, Kulbe H, Bellati F, Zizzari IG, Koshkaki HR, et al. Prognostic impact of aldehyde dehydrogenase-1 in ovarian cancer: a meta-analysis. Gynecol Oncol. 2018;150(1):151–159.
Chang B, Liu G, Xue F, Rosen DG, Xiao L, Wang X, et al. ALDH1 expression correlates with favorable prognosis in ovarian cancers. Mod Pathol. 2009;22(6):817–823.
Izycka N, Rucinski M, Andrzejewska M, Szubert S, Nowak-Markwitz E, Sterzynska K. Prognostic value of cancer stem cell marker expression for survival in ovarian cancer patients. Int J Mol Sci. 2023;24(3):2400.
Roque K, Heredia Vallejos MJ, Ruiz R, Galvez-Nino M, Flores CJ, Castro-Mollo MW, et al. Prognostic factors for survival in advanced epithelial ovarian cancer: a retrospective analysis.
Zong X, Nephew KP. Ovarian cancer stem cells: role in metastasis and opportunity for therapeutic targeting. Cancers (Basel). 2019;11(7):934.
Wilczyński JR, Wilczyński M, Paradowska E. Cancer stem cells in ovarian cancer—a source of tumor success and a challenging target for novel therapies. Int J Mol Sci. 2022;23(5):2496.
Alizadeh H, Akbarabadi P, Dadfar A, Tareh MR, Soltani B. Ovarian cancer stem cells: mechanisms and therapeutic opportunities. Mol Cancer. 2025;24(1):135.
Frąszczak K, Barczyński B. Role of cancer stem cell markers in ovarian cancer. Cancers (Basel). 2023;16(1):40.
Song P, Gao Z, Bao Y, Chen L, Huang Y, Liu Y, et al. Wnt/β-catenin signaling pathway in carcinogenesis and cancer therapy. J Hematol Oncol. 2024;17(1):46.
Martincuks A, Li PC, Zhao Q, Zhang C, Li YJ, Yu H, et al. CD44 in ovarian cancer progression and therapy resistance: a critical role for STAT3. Front Oncol. 2020;10:589601.
Yang L, Shi P, Zhao G, Xu J, Peng W, Zhang J, et al. Targeting cancer stem cell pathways for cancer therapy. Signal Transduct Target Ther. 2020;5(1):8.
Yang Y, Zhu G, Yang L, Yang Y. Targeting CD24 as a novel immunotherapy for solid cancers. Cell Commun Signal. 2023;21(1):312.
Zhao W, Zang C, Zhang T, Li J, Liu R, Feng F, et al. Prognostic value of ALDH1 in ovarian cancer: a meta-analysis. Onco Targets Ther. 2018;11:1821–1831.
Lee H, Kim B, Park J, Park S, Yoo G, Yum S, et al. Cancer stem cells: landscape, challenges and emerging therapeutic innovations. Signal Transduct Target Ther. 2025;10:1–18.
Ma H, Tian T, Cui Z. Targeting ovarian cancer stem cells: a new way out. Stem Cell Res Ther. 2023;14(1):28.
Kos M, Mertowska P, Mertowski S, Roliński J, Krasińska-Płachta A, Urbanowicz T, et al. From defense to disease: how the immune system fuels epithelial–mesenchymal transition in ovarian cancer. Int J Mol Sci. 2025;26.
BISAC
- MED058160 Medical / Nursing / Oncology & Cancer
Published
Series
Categories
License

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.







